
Our Process
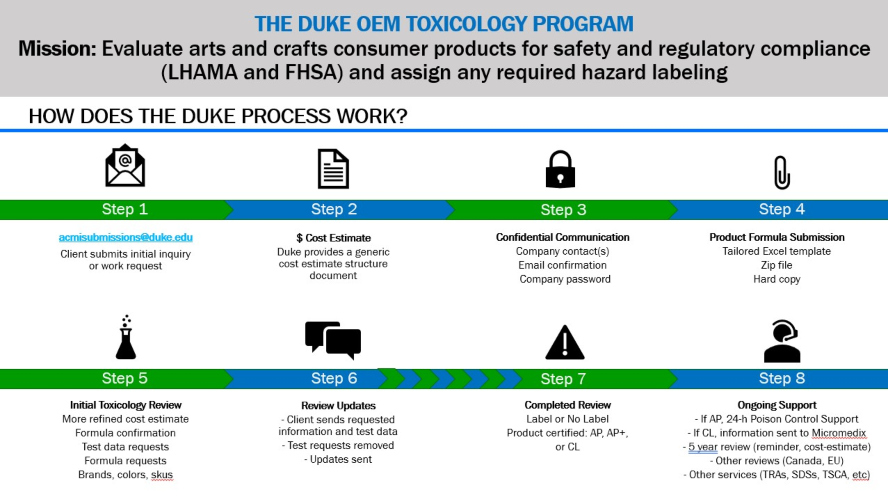
1. Client submits initial inquiry or work request
2. $ Cost Estimate
- Duke provides a generic cost estimate structure document
3. Confidential Communication
- Company contact(s)
- Email confirmation
- Company password
4. Product Formula Submission
- Tailored Excel template
- Zip file
- Hard copy
5. Initial Toxicology Review
- More refined cost estimate
- Formula confirmation
- Test data requests
- Formula requests
- Brands, colors, skus
6. Review Updates
- Client sends requested information and test data
- Test requests removed
- Updates sent
7. Completed Review
- Label or No Label
- Product certified: AP, AP+, or CL
8. Ongoing Support
- If AP, 24-h Poison Control Support
- If CL, information sent to Micromedix
- 5 year review (reminder, cost-estimate)
- Other reviews (Canada, EU)
- Other services (TRAs, SDSs, TSCA, etc)
Electronic Submissions
The Duke Toxicology Program has an Excel system in place for product formulation submissions for evaluation. If you would like to receive a copy of the Excel template, please email dukesubmissions@duke.edu.
Required Testing
Prior to the Duke Toxicology Program issuing an initial or renewed toxicologist’s approval for Certification by the Art and Creative Materials Institute (ACMI), certain testing may be required. Tests that may be required as part of the evaluation of your product and the corresponding test method can be found at the link below:
Duke has researched the following analytical laboratories that can perform these tests at different locations around the world. Please reach out to our administrative personnel (dukesubmissions@duke.edu) for more information.
- Eurofins – locations in the U.S, Europe, and Asia
- AM Testing – Florida location
- Applied Technical Services (ATS) – based in Atlanta
- Unitec –multiple locations in Asia
- Kirby – USA location only
- EMSL – Asbestos only (U.S locations)
Certification Standards
We evaluate the chronic hazard potential of consumer products against ASTM D4326, Labeling Art Materials for Chronic Health Hazards. This Standard has been codified in law as the Labeling for Hazardous Art Materials Act, which is regulated by CPSC. Test methods have been developed to support this Standard including measuring the extractability of metals from art materials (ASTM D5517-14), measuring the potential of consumer products to spontaneously combust (ASTM D6801-07 (2015)), and measuring whether or not aerosols present an aspiration risk (ASTM D7952-15). These standard test methods support the evaluation of consumer products both in the US and Canada.